Introduction
For most of us, sitting down to at least three meals a day is so normal that intentionally refusing to eat seems unthinkable. Yet for some, the experience of eating is fraught with anxiety. Every moment of every day is preoccupied with thoughts of food, and the fear of gaining weight is so intense that giving in to hunger is unfathomable. This is the reality for people who suffer from anorexia nervosa, commonly referred to as anorexia, a mental illness categorized as a feeding and eating disorder.
Background
Although the development of anorexia is hard to pinpoint, signs of the disorder in patients have become increasingly recognizable, and a standard has been set for diagnosis. The most recent Diagnostic and Statistical Manual of Mental Disorders (DSM–5) provides three criteria for a diagnosis of anorexia nervosa [1]. First, the patient must be engaging in a restriction of energy intake—meaning food—that is below what they need to maintain a healthy body weight, leading to a less than minimally normal body weight for their age, sex, and physical health. The current index used to determine healthy versus unhealthy body weight is the body mass index or BMI. Second, the patient must display an intense fear of gaining weight or persistent behavior that interferes with weight gain. Fear is often difficult to quantify, so visible symptoms such as food avoidance, excessive exercise, and other activities that prevent weight gain are easier to accurately measure for the diagnostic process. Finally, the patient must overly consider body weight or shape during self-evaluation, deny the medical seriousness of low body weight, or show a disturbance in the way in which their body weight or shape is experienced. This disturbance is defined as the inability for the patient to recognize they are at a low weight, often paired with the insistence that they are at a healthy or even high body weight despite evidence to the contrary. Similar to the second criterion, these symptoms are often difficult to identify when relying solely on patient disclosure. When all three of these symptoms are present, the patient is diagnosed with anorexia nervosa [1]. Alternatively, when a patient displays all the features of anorexia but lacks the severe low body weight, they are diagnosed with atypical anorexia. Patients with atypical anorexia can have just as severe medical and psychiatric complications as patients with anorexia [2].
The causes of anorexia nervosa are still largely unknown, although a combination of biological, psychological, and social components are understood as contributing factors [2]. Biological factors like genetics play a significant role in the likelihood of developing anorexia nervosa. For example, abnormalities in neurotransmitters, the chemical messengers of the nervous system, are also associated with anorexia nervosa. Psychologically, certain personality traits are commonly associated amongst people who suffer from anorexia nervosa—poor distress tolerance, perfectionism, neuroticism, obsessionality, harm avoidance, and inflexibility correlate with this disorder [3]. Anorexia is also closely connected to anxiety, as over two-thirds of patients are also diagnosed with an anxiety disorder [4]. Finally, social and external environmental factors also impact the risk of developing anorexia nervosa. Stressful life transitions, such as going to college, moving, or losing a parent, as well as forms of bullying, teasing, or abuse, are thought to be potential contributing factors to anorexia. Notably, dieting is a strong risk factor for anorexia nervosa [5].
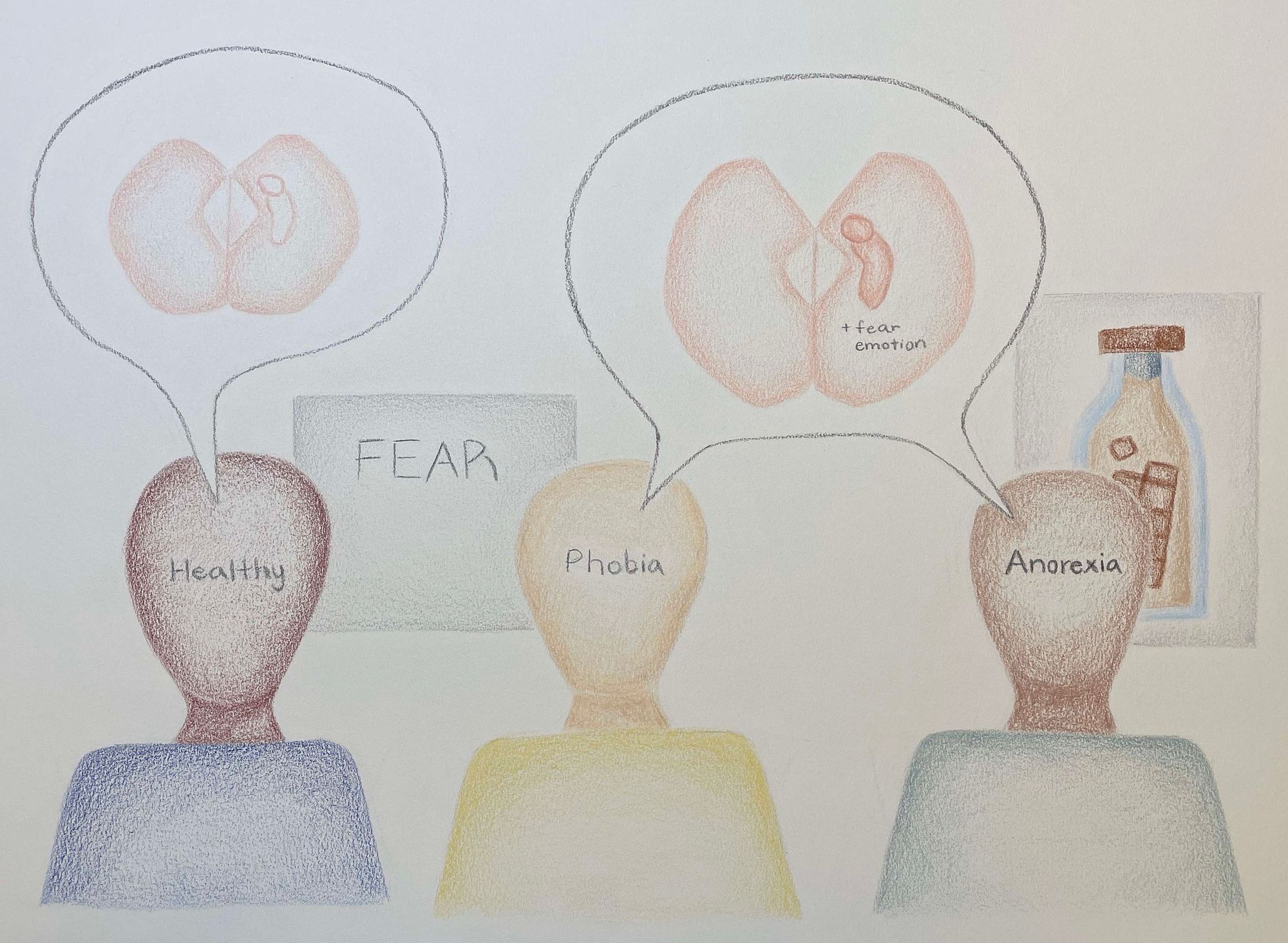
As the diagnostic criteria make clear, anorexia manifests in a severe reluctance to gain weight to the point where individuals cause damage to their physical health. The prioritization of this goal leads to changes in the perception of food and taste-related stimuli. For example, when shown pictures of high-calorie drinks, patients with anorexia displayed activity in the left amygdala and hippocampal region of the brain. These areas are associated with positive and negative emotional responses, the experience of fear, and the encoding of verbal and visual-spatial memories [6, 7]. This data was collected through magnetic-resonance imaging (MRI) in female patients during acute phases of anorexia and in healthy patients of the same age. In this study, the neural response exhibited by individuals with anorexia mirrored that of individuals who confronted their phobias whereas healthy controls exhibited a neutral or pleasurable response [7]. Because of this strong fearful emotional response, researchers believe that anorexia creates an aversive perception of high-calorie foods, stimuli to which individuals without anorexia see in a neutral or pleasurable light.
Researchers believe these changes in response to pleasurable food stimuli indicate “reward contamination”, where the notion of what normally serves as rewarding is flipped in someone with anorexia [8]. For instance, palatable food and taste stimuli evoke fear and aversion in a patient with anorexia, where unpleasurable activities, like food restriction, become positive and rewarding. Restrictive behaviors can also be negatively enforced. Increased body dissatisfaction, a trait universal in all cases of anorexia, can result in negative feelings and thought patterns related to eating. When eating is perceived as a negative behavior, self-starvation becomes a positive response designed to lessen the mental anguish felt by eating [8]. Paired with changes in response to food stimuli, this further reinforces the sense of reward contamination experienced by individuals with anorexia. Furthermore, when shown pictures of aversive food stimuli, like moldy strawberries, individuals with anorexia displayed increased activity in the anterior cingulate cortex (ACC) [9]. The ACC is a region of the brain involved in emotion assessment, emotion-related learning, and emotional regulation, and reward and punishment processing [10]. Patients with anorexia displayed significantly more sensitive reactions to both positive and negative food stimuli than healthy controls. The activation of the ACC in response to negative food stimuli alongside the aforementioned changes in response to pleasurable food stimuli shows a heightened neural response to all food cues in patients with anorexia. This indicates that individuals with anorexia place disproportionate emphasis on evaluating food and taste-related stimuli, which lays the foundation for more sensitive reward responses relating to food [11].
Dopamine is a neurotransmitter that plays a major role in abnormalities of motivation and reward related to the restrictive eating patterns found in anorexia [2]. So how exactly is dopamine connected to reward pathways? Dopamine is involved in three separate functions of the brain’s reward response. First, it is linked to the pleasure impact of rewards, referred to as “liking” something. Second, it is linked to the pursuit of reward through motivation or incentivization of stimuli resulting in reward, referred to as “wanting” something. Lastly, it is linked to the learned associations of reward stimuli to the feeling of reward itself, referred to as “learning” [12]. Anorexia impacts each of these three stages of the reward system. Just as how the disorder leads to an aversive perspective towards high-calorie food stimuli because of its association with weight gain, it amplifies the value of behaviors that fit the ultimate goal of weight loss, intensifying the “liking” and “wanting” responses to actions such as food restriction or excessive exercise [13]. The increased pleasure response evoked from cues and behaviors previously noted reinforces the pursuit of weight loss as a rewarding experience. This is an example of habit forming, which is the third element of the reward system. As the individual learns that restrictive behaviors will provide a consistent reward response, they begin to take on habitual cognitive biases in favor of actions like self-starvation.
Dopaminergic neurons are known to be involved with reward, yet they are also activated during experiences of physical and emotional stress [2]. Severely restrictive dieting, paired with excessive exercise, can lead to a stress response in the body. In response to that stress, specific neurons release a flood of dopamine. This consistent elevated dopamine release that occurs during the dieting routines typical of anorexia is thought to create a reward pathway that conditions the brain to value self-starvation [14]. The dopamine response is particularly high because dopamine plays a critical evolutionary role in facilitating foraging for food [15]. When food intake is suppressed, whether intentionally or unintentionally, dopamine responses increase to motivate food-seeking behavior. The dopamine released from stress is combined with the dopamine already released for reward, causing an additive effect. As a result, the chronic stress produced by self-starvation amplifies the value of this behavior by making the individual more sensitive to the dopamine reward system. This further amplifies the value of behaviors associated with meeting the weight loss goal [14]. Because dopamine increases can be a cause or result of food restriction, these neural changes may play no causative role in anorexia, but rather act purely as a symptom of it. Therefore it is difficult to determine whether changes to the dopamine pathway are a cause of anorexia or a result of the starvation behaviors perpetuated by the disorder.
Researchers have also found that the altered dopamine status of patients with anorexia may result from a brain abnormality which underlies the disorder. Disturbances in dopamine metabolism may arise from genetic factors rather than the engagement in restrictive behaviors. [15]. Researchers at Cambridge found that genetics and non-shared environmental factors contribute anywhere from 48 to 74% of the risk for developing anorexia nervosa. This data was collected through a twin study, where 672 pairs of female 17 year-old twins were assessed for anorexia nervosa [16]. Twin studies are particularly useful for genetic observation because the identical genetic makeup of twins allows scientists to identify differences between genetic and environmental influences on behavior.
In a different study, researchers wanted to assess if genetic changes to the dopamine pathway may impact susceptibility to developing anorexia [17]. In this study, the researchers used two strains of mice: one which had genetic changes that made it resistant to obesity when fed a high-fat diet, and one which was genetically identical to the first strain in all ways except resistance to obesity. The resistant strain is often used as a model for anorexia among researchers. The mice were then placed on a restricted diet, where researchers would restrict their access to food to two hours a day for five days, and provided with an exercise wheel. The nonresistant mice responded with normal behaviors to a lowered meal plan and exercise access: they displayed lowered exercise wheel use and higher anticipatory behaviors around feeding time. The other strain, however, developed signs of anorexia and displayed alteration in dopaminergic pathways. Particularly, the striatum, a brain region associated with decision-making, motivation, reinforcement, and reward perception, showed upregulation of dopamine receptors in these mice. Here, upregulation is a process where the number of dopamine receptors increases, resulting in more sensitivity and an increased response to certain stimuli, like restricted access to food. In the mice where this change was present, the development of weight loss behaviors, such as increased running wheel activity and lack of anticipatory actions before feeding time, occurred. Thus, brain changes relating to dopamine stemming at some level from genetic factors may play a more causative role in the development of anorexia [17].
Ultimately, dopamine functions within anorexia to attach positive feelings of reward to restrictive behaviors. The responses associated with maladaptive behaviors are perceived as pleasurable in individuals with anorexia. This pleasurable or rewarding feeling is what helps create habitual routines of these maladaptive behaviors. This reward-based learned behavior is similar to the brain patterns that are produced by drug addiction. Particularly, the role of dopamine-associated behaviors in both anorexia and addiction supports the understanding that a common neural mechanism exists in both the reinforcing values of restrictive eating behaviors as well as behaviors in drug addictions [12]. The intensity of the dieting and exercise routines common within anorexia parallel the clinical profile of addiction, where individuals become preoccupied with excessive drug-seeking and drug-use behaviors [18]. Individuals with anorexia and individuals with addiction display a similar loss of control over symptoms as well as denial of the extent to which the disorder impacts their lives, and both continue to pursue unhealthy goals—weight loss and drug abuse—despite adverse physical, emotional, and social consequences [1]. Additionally, both conditions have high mortality rates and high rates of relapse, or worsening of symptoms following some improvement [19]. However, anorexia is a complex disease, and while it shares similarities with addiction, the two are not identical in many facets.
One primary overlap between the two is that brain regions impacted by drug addiction also show similar developments in anorexia. With addiction, changes in the striatum result in the creation of additional dopamine receptors following the onset of substance-use behaviors [20]. These additional receptors increase sensitization to stimuli that produce rewarding feelings--in the case of addiction, the stimulus is the drug itself. As this sensitivity continues to increase, with more and more dopamine receptors required to create a sufficiently rewarding response, the brain’s perception of what is rewarding becomes narrowed [20]. Once this shift occurs, no other stimuli can parallel the intense and almost immediate reward produced by drugs, reinforcing the addiction behavior even at the cost of relationships and activities that once held significant value [21]. This same pattern occurs nearly step-for-step in anorexia. When the goal of weight loss becomes prioritized over all else, actions that reinforce that goal such as restrictive eating patterns, excessive exercise, and abuse of laxatives result in rewarding feelings. As sensitivity to the dopamine pathway increases, individuals with anorexia begin to engage in more and more restrictive behaviors [13]. As this cycle intensifies, individuals prioritize restrictive behaviors over all else, even elements of their life they once found rewarding. Just like how the rewarding feelings resulting from drug use make treating addiction difficult, these physiological and psychological effects produced by anorexia make many patients resistant to treatment.
So why is anorexia-related research important? Given how anorexia’s effects can produce resistance to treatment, combined with the poorly understood origin and development of the disorder, anorexia nervosa is extremely difficult to treat [22]. On average, less than half of patients recover, with one-third of patients alternating between states of partial remission and relapse and 20% remaining chronically ill even 21 years after the onset of the disorder [2]. Alongside this grim prognosis, anorexia nervosa is widely recognized as having the highest mortality rate of all mental health disorders, which stems from starvation-related complications as well as suicide [23]. Furthermore, specialized treatment is often expensive, difficult to access, and so terrifying for many patients that they refuse it. In fact, 20–51% of inpatients and 23–73% of outpatients avoid or leave their treatment program [24]. Additionally, only approximately 50% of anorexia nervosa cases receive an official diagnosis and only 1 in 3 of those diagnosed receive specialized care [25]. That means countless individuals are struggling with this life-threatening disorder without any form of treatment, let alone specialized care.
Even so, there is hope for a brighter future. Developments in studying the neurobiological origins of anorexia nervosa may provide a path forward for better, more effective treatment and prevention strategies. There is a need to better understand the role of dopamine in the progression and development of anorexia. Such progression would allow for potential use of dopaminergic manipulation strategies for improvement of treatment. Studies that delve more specifically into the role of dopamine in anorexia will almost certainly shed light on the neurobiological underpinnings of the disorder. Furthermore, while plenty of research exists for the role of dopamine reward systems in drug addiction, the same cannot be said for anorexia. Engaging in this type of research will not only benefit the treatment and prevention of anorexia but provide a more thorough and nuanced understanding for providers treating drug addiction and other reward-based disorders. It is imperative that researchers pursue this type of inquiry, as current treatment options are not entirely effective in saving the lives of people struggling with severe anorexia.
References
1. American Psychiatric Association. (2013). Diagnostic and statistical manual of mental disorders (5th ed.). Washington, DC.
2. Moskowitz L, Weiselberg E. Anorexia Nervosa/Atypical Anorexia Nervosa. Curr Probl Pediatr Adolesc Health Care. 2017 Apr;47(4):70-84. doi: 10.1016/j.cppeds.2017.02.003. PMID: 28532965.
3. The Price Foundation Collaborative Group. (2001). Deriving behavioural phenotypes in an international, multi-centre study of eating disorders. Psychological Medicine, 31(4), 635-645. doi:10.1017/S0033291701003671.
4. R.A. Sansone, L.A. Sansone. Personality pathology and its influence on eating disorders. Innovat Clin Neurosci, 8 (2011), pp. 14-18.
5. E. Stice. Risk and maintenance factors for eating pathology: a meta-analytic review. Psychol Bull, 128 (2002), pp. 825-848.
6. Z. Ellison, J. Foong, R. Howard, E. Bullmore, S. Williams, J. Treasure. Functional anatomy of calorie fear in anorexia nervosa. Lancet, 352 (1998), p. 1192.
7. K.R. Kim, J. Ku, J.H. Lee, H. Lee, Y.C. Jung. Functional and effective connectivity of anterior insula in anorexia nervosa and bulimia nervosa. Neurosci. Lett., 521 (2012), pp. 152-157.
8. C.G. Fairburn, Z. Cooper, R. Shafran. Cognitive behaviour therapy for eating disorders: a transdiagnostic theory and treatment. Behav. Res. Ther., 41 (2003), pp. 509-528.
9. F.A. Cowdrey, R.J. Park, C.J. Harmer, C. McCabe. Increased neural processing of rewarding and aversive food stimuli in recovered anorexia nervosa. Biol. Psychiatry, 70 (2011), pp. 736-743.
10. M.M. Botvinick. Conflict monitoring and decision making: reconciling two perspectives on anterior cingulate function. Cogn. Affect. Behav. Neurosci., 7 (2007), pp. 356-366.
11. C. Keating. Theoretical perspective on anorexia nervosa: the conflict of reward. Neurosci. Biobehav. Rev., 34 (2010), pp. 73-79.
12. K.C. Berridge, T.E. Robinson, J.W. Aldridge. Dissecting components of reward: ‘liking’, ‘wanting’, and learning. Curr. Opin. Pharmacol., 9 (2009), pp. 65-73.
13. Caitlin B. O’Hara, Iain C. Campbell, Ulrike Schmidt. A reward-centred model of anorexia nervosa: A focussed narrative review of the neurological and psychophysiological literature, Neuroscience & Biobehavioral Reviews, Volume 52, 2015, Pages 131-152, ISSN 0149-7634, https://doi.org/10.1016/j.neubiorev.2015.02.012.
14. Södersten P, Bergh C, Leon M, Zandian M. Dopamine and anorexia nervosa. Neurosci Biobehav Rev. 2016 Jan;60:26-30. doi: 10.1016/j.neubiorev.2015.11.003. Epub 2015 Nov 19. PMID: 26608248.
15. Kaye W. Neurobiology of anorexia and bulimia nervosa. Physiol Behav. 2008 Apr 22;94(1):121-35. doi: 10.1016/j.physbeh.2007.11.037. Epub 2007 Nov 29. PMID: 18164737; PMCID: PMC2601682.
16. K.L. Klump, K.B. Miller, P.K. Keel, M. McGue, W.G. Iacono. Genetic and environmental influences on anorexia nervosa syndromes in a population-based twin sample. Psychol Med, 31 (2001), pp. 737-740 (GENES).
17. C. Gelegen, J. Van Den Heuvel, D.A. Collier, I.C. Campbell, H. Oppelaar, E. Hessel, M.J.H. Kas. Dopaminergic and brain-derived neurotrophic factor signalling in inbred mice exposed to a restricted feeding schedule. Genes Brain Behav., 7 (2008), pp. 552-559.
18. G.F. Koob, N.D. Volkow. Neurocircuitry of addiction. Neuropsychopharmacology, 35 (2010), pp. 217-238.
19. N.D. Berkman, K.N. Lohr, C.M. Bulik. Outcomes of eating disorders: a systematic review of the literature. Int. J. Eat. Disord., 40 (2007), pp. 293-309.
20. Marsch, L.A., and W.K. Bickel. 2001. Toward a behavioral economic understanding of drug dependence: delay discounting processes. Addiction. Doi: 10.1046/j.1360-0443.2001.961736.x.
21. Lewis, Marc C. “Addiction and the Brain: Development, Not Disease.” Neuroethics, vol. 10, no. 1, 11 Jan. 2017, pp. 7–18.
22. Steinglass JE, Berner LA, Attia E. Cognitive Neuroscience of Eating Disorders. Psychiatr Clin North Am. 2019 Mar;42(1):75-91. doi: 10.1016/j.psc.2018.10.008. Epub 2018 Dec 3. PMID: 30704641; PMCID: PMC6601331.
23. Jagielska G, Kacperska I. Outcome, comorbidity and prognosis in anorexia nervosa. Psychiatr Pol. 2017 Apr 30;51(2):205-218. English, Polish. doi: 10.12740/PP/64580. Epub 2017 Apr 30. PMID: 28581532.
24. Abbate-Daga G, Amianto F, Delsedime N, De-Bacco C, Fassino S. Resistance to treatment and change in anorexia nervosa: a clinical overview. BMC Psychiatry 2013; 13: 294–312.
25. Keski-Rakhonen A, Hoek HW, Susser ES, Linna MS, Sihvola E, Raevuori A et al. Epidemiology and course of anorexia nervosa in the community. Am. J. Psychiat. 2007; 164: 1259–1265.
