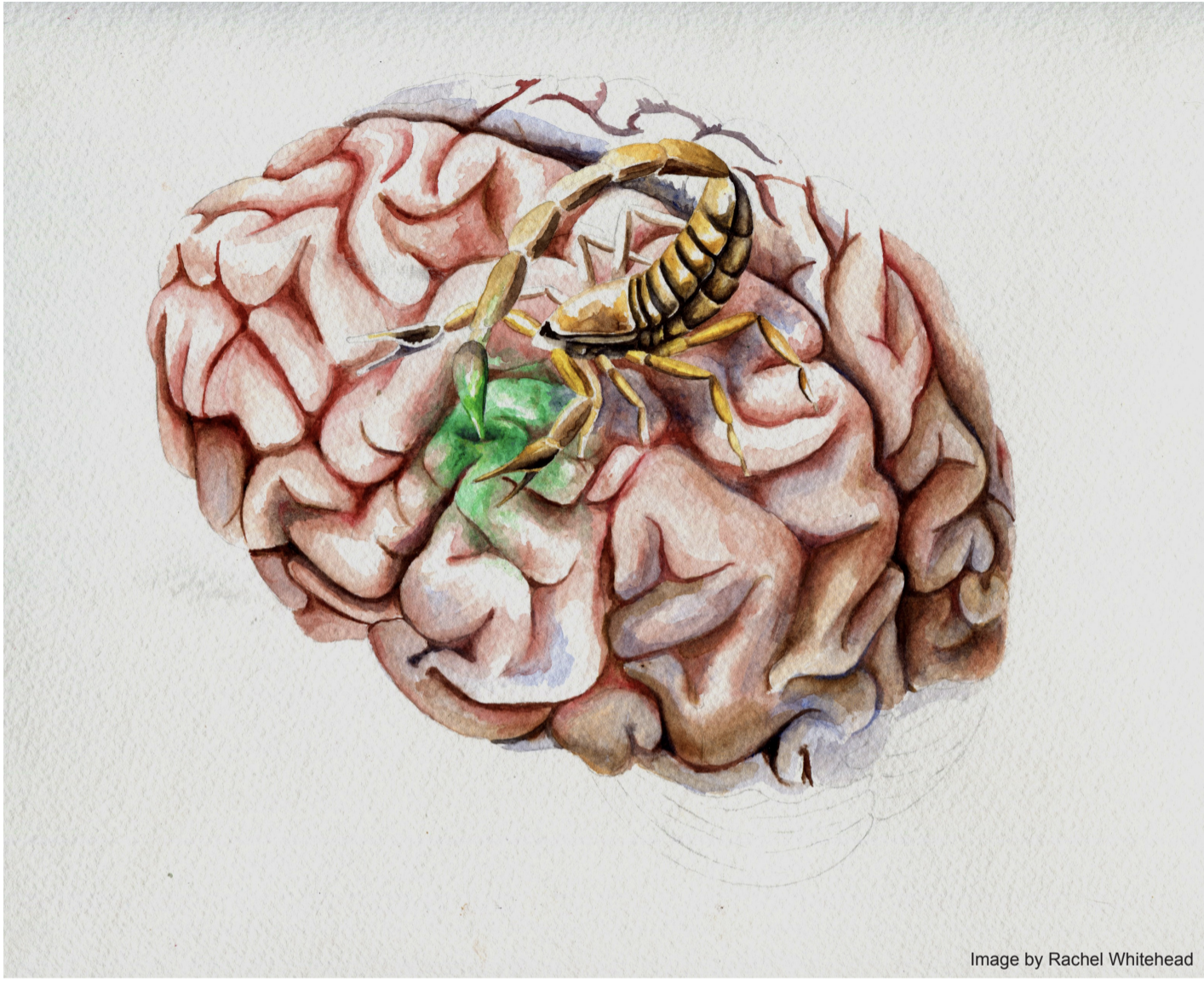
In 2010, Dr. Jim Olson founded the Seattle-based company Blaze Bioscience to test and develop Tumor Paint for clinical use. Tumor Paint BLZ-100, Blaze Bioescience’s first candidate, is currently undergoing clinical trials in Australia.
This first round of testing includes adult patients with non-metastatic skin cancers to determine the safety and potential toxicity of intravenous injection of the compound. Phase 1 of this trial, which is currently ongoing, is designed to determine safe levels of the compound in patients. Phase 2 will consist of pharmokinetic studies of the compound in the human body, as well as fluorescence studies in resected cancerous tissue. In this part of the trial, patients will be injected with an initial dose of BLZ-100 before surgery, then placed into five different treatment groups and administered varying levels of the compound during the procedure. Clinicians will test for adverse effects directly after injection, as well as 3, 5, and 8 days after injection [1].
In September 2013 the United States Food and Drug Administration approved Phase 1 Investigational New Drug clinical trials in patients with grade I, II, III, and IV gliomas intended for surgical removal [2]. This study is currently accepting candidates, although Blaze Bioscience does not believe that BLZ-100 will be available for commercial use until 2020 [3].
Since the discovery of Tumor Paint, many have wondered if the tumor-binding properties of Chlorotoxin could be combined with a tumor-killing agent to find and destroy any solid cancerous tumor in the body. Olson’s current research, and the future vision of Blaze Bioscience, is to identify and isolate specific components of compounds produced by plants and animals that will selectively bind to cancerous cells. Project Violet, a crowd-funded initiative, hopes to identify, develop, and engineer optimized peptides, known as optides, for the detection and treatment of malignant tissue [4].